Refine by
Implant Tissue Equipment & Supplies In Grenada
46 equipment items found
Manufactured by:EnColl Corporation based inFremont, CALIFORNIA (USA)
Surgicoll Mesh is a biocompatible collagen mesh that is flexible, strong yet soft. The collagen membrane is also supple when hydrated and handles like natural tissue. It readily conforms to the surgical site upon application and is easily sutured. Surgicoll- Mesh is constituted by Type-I collagen membrane that is free of contaminants like lipids, elastin and other immunogenic proteins. The ...
Manufactured by:Cook Biotech Inc. based inWest Lafayette, INDIANA (USA)
The Biodesign Anal Fistula Plug is for implantation to reinforce soft tissue where a rolled configuration is required, for repair of anorectal ...
Manufactured by:Cook Biotech Inc. based inWest Lafayette, INDIANA (USA)
The Biodesign Enterocutaneous Fistula Plug is for implantation to reinforce soft tissue for repair of enterocutaneous ...
Manufactured by:Cook Biotech Inc. based inWest Lafayette, INDIANA (USA)
The Biodesign Fistula Plug is for implantation to reinforce soft tissue for repair of recto-vaginal or anorectal ...
Manufactured by:Cook Biotech Inc. based inWest Lafayette, INDIANA (USA)
The Biodesign Hernia Graft is intended for implantation to reinforce soft tissues where weakness exists. Indications for use include the repair of a hernia or body wall ...
Manufactured by:Cook Biotech Inc. based inWest Lafayette, INDIANA (USA)
The Biodesign Hiatal Hernia Graft is used for implantation to reinforce soft tissue where weakness exists, including repair of hiatal ...
Manufactured by:USIOL. Inc. based inLexington, KENTUCKY (USA)
Second generation, high molecular weight, high viscosity ophthalmic gel. Maintains ocular surgical space and prevents leakage from capsule opening. Lubricates critical tissue, implant and instruments for ease of manipulation and insertion. Easily removed following lens implantation. Isotonic, non-pyrogenic and noninflammatory with excellent flow ...
Manufactured by:Cook Biotech Inc. based inWest Lafayette, INDIANA (USA)
The Biodesign Peyronie’s Repair Graft is intended for implantation to reinforce soft tissue where weakness exists in the urological anatomy, including but not limited to repair of tunica albuginea defects and reinforcement in the repair of Peyronie’s ...
Manufactured by:BioIntegral Surgical Inc. based inMississauga, ONTARIO (CANADA)
No reports of dilatation, insufficiency, tissue failure, thromboembolism, or bleeding events, No reports of calcification in 18 years and 13 000 implants, No evidence of tissue degeneration ...
Manufactured by:Crossroads Extremity Systems based inMemphis, TENNESSEE (USA)
There are potential risks associated with the use of these devices, some of which include: allergic reaction to the implant material, fracture of the implant, soft-tissue complication (e.g., infection at the implant site, prolonged healing), and revision surgery. Refer to IFU for all contraindications, warnings, and risks. ...
Manufactured by:Betatech Medical based inISTANBUL, TURKEY
Betamix Vaginal Sling is a mesh implant for reinforcing connective tissue structures and ligaments. Betamix is knitted from non-resorbable, monofilament polypropylene fibre. The specially developed knitted structures and ideal mesh size of Betamix are optimally matched to the fields of application for which it is ...
Manufactured by:Vulkan Implants based inBarcelona, SPAIN
Designed to increase primary stability and peri-implant crestal bone tissue stability. An ideal system for immediate post-extraction implantation, for immediate loading and offering new prosthetic solutions focused on soft tissue ...
Manufactured by:OSARTIS GmbH based inMünster, GERMANY
BonOs® R NF and BonOs® R NF Genta are fast-setting acrylic bone cements for use in bone surgery. BonOs® R NF Genta contains the antibiotic gentamicin sulfate that protects the implant and the surrounding tissue from colonization with pathogens that are sensitive to ...
Manufactured by:Anthogyr - Institut Straumann AG based inSallanches, FRANCE
Axiom® Tissue Level implant promotes biological safety and easy prosthesis insertion at the gingival level. It helps to preserve the epithelium and connective tissue attachment. The Axiom® TL implant is available in the REG profile, suitable for most clinical indications, and PX profile preferred for immediate ...
Manufactured by:Anthogyr - Institut Straumann AG based inSallanches, FRANCE
Axiom® Multi Level® is an implant concept that offers a smart range of surgical solutions and complementary philosophies between Bone and Tissue Level implants. Axiom® Multi Level® philosophy is fully driven by prosthetics outcome for better patients satisfaction, with a strong orientation for digital and CAD/CAM custom-made ...
Manufactured by:MBP – Medical Biomaterial Products GmbH based inNeustadt-Glewe, GERMANY
Rematrix is a cell-free dermal collagen matrix derived from porcine dermis (ADM). Rematrix is intended for use as a surgical biomaterial implant for soft tissue repair, serving as a scaffold material, tissue coverage or tissue replacement. The double sterile packed membrane is available in various sizes. Rematrix is sold in dry ...
Manufactured by:Novus Life Sciences based inFotan, HONG KONG
It presents the added advantage of bioactivity, which facilitates the bone healing process, rendering it highly suitable for permanent medical implant applications and bone tissue engineering ...
Manufactured by:Beijing Biosis Healing Biological Technology Co., Ltd. based inBeijing, CHINA
This membrane features a natural fibrous porosity and is abundant in bioactive substances, enhancing its biological inductive activity. It facilitates tissue regeneration by promoting rapid vascularization, cell differentiation, and mucosal crawling. The membrane integrates seamlessly with host tissues upon implantation, ensuring bioactivity ...
Manufactured by:Heraeus Medical GmbH based inWehrheim, GERMANY
The simultaneous release of gentamicin and clindamycin addresses up to 90% of pathogens found in prosthetic joint infections (PJI). It provides an initial high local antibiotic output to prevent bacterial colonization on implants and adjacent tissue. This antibiotic-loaded cement is particularly beneficial for high-risk patients undergoing primary arthroplasty, ...
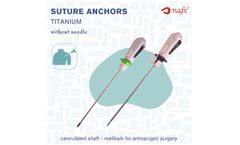
Manufactured by:Nafx Medical based inCankaya, TURKEY
Suture Anchor With Cannulated Shaft - Without Preloaded Needle is a single-use orthopedic implant designed for soft tissue fixation in arthroscopic and open procedures. Featuring a cannulated shaft, it allows precise placement over a guide wire and facilitates bone tunnel access. This anchor is optimized for manual suture loading, offering flexibility in suture ...