- Home
- Equipment
- usa illinois
- bone replacement
Show results for
Refine by
Bone Replacement Equipment & Supplies In Illinois
28 equipment items found
Manufactured by:Brasseler USA based inSavannah, GEORGIA (US) (USA)
Designed and manufactured to very rigid specifications using only the finest surgical grade stainless steel. Brasseler USA Surgical Instrumentation is pleased to introduce our Large Bone Saw Blade Replacement line for Original Equipment Manufacturers (OEMs) Saw Blades. Our comprehensive line of high-quality replacement surgical large ...
Manufactured by:Parametrics Medical based inLeander, TEXAS (USA)
OsteoSource™ allograft bone particulates have osteoconductive properties and are well suited to fill bone structure defects. Osteoconductive allografts act as a scaffold for the growth of natural bone. Over time, the natural bone replaces the donor’s bone. OsteoSource™ ...
Manufactured by:Bonegraft Biyolojik Malzemeler San. VE TIC. A.S. based inIZMIR, TURKEY
Bone Cement based on polymethyl methacrylate (PMMA) is a widely used biomaterial due to its ease of use in clinical practice and especially the long survival rate proven by dentures. Common Indications for Bone Cement: total joint replacement are bone and joint reconstructions, fracture fixation and treatment of osteoporotic ...
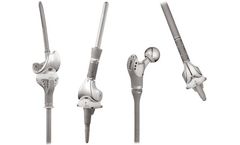
Manufactured by:Microport Orthopedics Inc. based inArlington, TENNESSEE (USA)
The Profemur® line of primary hip stems has an established clinical history. Each Profemur® hip stem offers its own philosophy to provide fixation and replace bone anatomy. Triple taper stem philosophy" Designed for primary hip replacement surgery, the Profemur® Gladiator® hip stem is a triple tapered wedge stem with a distinct ...
Manufactured by:Zimmer Biomet based inWarsaw, INDIANA (USA)
Bonus Synthetic Bone Graft Substitute granules are resorbable, osteoconductive matrices consisting of a thin, 2–10 micron layer of hydroxyapatite over a calcium carbonate core. Bonus Synthetic Bone Graft Substitute has been indicated as a bone graft substitute that resorbs and is replaced with bone during ...
by:Gain Therapeutics, Inc. based inBethesda, MARYLAND (USA)
These mutations result in the misfolding and subsequent dysfunction of GLB, which leads to the toxic substrate accumulation of keratan sulfate in organs and tissues. Very limited and investigational symptomatic treatment options include substrate reduction therapy, enzyme replacement therapy, bone marrow transplantation, stem cell transplantation and gene therapy ...
by:Gain Therapeutics, Inc. based inBethesda, MARYLAND (USA)
These mutations result in the misfolding and subsequent dysfunction of GLB, which leads to the toxic substrate accumulation of GM1 ganglioside in organs and tissues. Very limited and investigational symptomatic treatment options include substrate reduction therapy, enzyme replacement therapy, bone marrow transplantation, stem cell transplantation and gene therapy ...
Manufactured by:Himed based inOld Bethpage, NEW YORK (USA)
Beta-tricalcium phosphate (β-TCP) is a calcium phosphate biomaterial noted for its bioactive and resorbable properties, which make it highly effective in applications such as bone grafting and implants. It supports bone regeneration due to its osteoconductive nature, acting as a scaffold for natural bone to replace over ...
Manufactured by:Medosis based inSofia, BULGARIA
Biongraft Flex is an advanced bone graft substitute designed primarily for dental applications, comprising beta-tricalcium phosphate (b-TCP) and a resorbable PLA-based synthetic polymer. This material gradually degrades to harmless byproducts, specifically carbon dioxide and water, facilitating a seamless integration with human physiology. The micro and macro porous architecture ...
Manufactured by:EOS MEDICAL SOLUTIONS (EOSMED) based inSofia, BULGARIA
Biongraft is an injectable and formable paste bone graft based on hydrogel and Beta-tricalcium phosphate (β-TCP), including ZrO2 nanoparticles for antibacterial e?cacy. Biongraft is a safe and fully biocompatible material which is designed to act as an osteoconductive sca?old to support the ingrowth and fusion of adjacent viable bone when placed in an ...
Manufactured by:Meticuly Co., Ltd. based inPathumwan District, THAILAND
Our patient-specific Mid-face Reconstruction Mesh is a patient-specific titanium mesh implant serving to replace defective bone lost in the cranio-maxillofacial region from trauma, congenital condition, or tumors. The implant’s contour is designed anatomically, based on CT scan data, to cover the bony void with overlapping margin. The implant has inbuilt ...
by:In2Bones Global based inMemphis, TENNESSEE (USA)
CoLink® Bone Graft Harvester is available in two Outer Tube diameters: 6mm and 8mm. ...
Manufactured by:United Orthopedic Corporation (UOC) USA Inc. based inIrvine, CALIFORNIA (USA)
vThe USTAR II System is designed for extensive reconstruction of the hip and knee joint. Components have been designed based on 20 years experience with the previous USTAR system. The modularity of the USTAR II System is designed to allow flexibility to address a variety of difficult surgical ...
Manufactured by:Synergy Biomedical, LLC based inWayne, PENNSYLVANIA (USA)
The 45S5 formulation of bioactive glass has been the primary material used in today's bioactive glass bone graft products (45% SiO2, 24.5% CaO, 24.5% Na2O, and 6% P2O5). ...
Manufactured by:OrthoProMed Inc. based inCovina, CALIFORNIA (USA)
Orthopromed, Inc. offers a specialized bone cement designed for joint replacement surgeries. This product is particularly beneficial for patients with allergies or sensitivities as it does not contain gentamicin, an antibiotic usually added to bone cements. It ensures strong adhesion, contributing to long-lasting surgical results. ...
Manufactured by:Parametrics Medical based inLeander, TEXAS (USA)
EnduriFuse is allograft bone containing viable bone-derived cells. This innovative graft contains properties ideal for the growth of natural bone: An osteoconductive three-dimensional scaffold with cortical and cancellous components. A demineralized bone scaffold with osteoinductive potential containing growth factors that recruit immature cells and stimulate those cells to develop into active ...
Manufactured by:Inion Oy based inTampere, FINLAND
Inion BioRestore™ is a synthetic bone graft substitute, which remodels into bone and is easy to use. Inion BioRestore™ is made of degradable bioactive glass, which once in contact with the natural body fluids forms a silica gel and calcium phosphate layer, providing scaffolding for the new bone tissue to be ...
Manufactured by:Parametrics Medical based inLeander, TEXAS (USA)
Machined interbody spacers provide a natural osteoconductive option for spine fusion. The allograft bone is precision machined to provide an optimal fit and stability between the allograft and adjacent vertebral bodies. Parametrics Medical offers spacers in various profiles, footprints, and ...
Manufactured by:Biogennix based inIrvine, CALIFORNIA (USA)
Agilon Moldable bone grafting product is the latest moldable grafting solution in Biogennix’ advanced bone graft product line. Developed with Biogennix proprietary TrelCor® technology, Agilon Moldable consists of a suspension of 1-2mm TrelCor granules in a rapidly absorbing, organic binder blended with type-1 ...
by:Cytex Therapeutics, Inc. based inDurham, NORTH CAROLINA (USA)
The cornerstone of Cytex technology is our patented approach to producing 3-Dimensional woven implants that mimic the natural function and performance of the cartilage that lines joints such as the hip, shoulder and knee. Cytex implants can be made to fit the complex curvature found in joints, thereby helping restore and maintain natural joint ...