- Home
- Equipment
- canada saskatchewan
- cell therapeutic
Refine by
Applications
- IDCT for Lumbar Disc Degeneration
- Therapy Discogenic Cell for Degenerative Disc Disease
- Medical solutions for MiniTC and mini-stem system sector
- Potential Functional Cures for Patients with Chronic Diseases
- Shielded Living Therapeutics Cell for Lysosomal Diseases
- Shielded Living Therapeutics Cell for Diabetes
- Shielded Living Therapeutics Cell for Rare Blood Disorders
- Solution for Clinical Trials
- Solution for Access to Investigational Medicines
- Software Solutions for Deep Mutational Scanning & Selection
- Software Solutions for COVID-19
Cell Therapeutic Equipment & Supplies Near Saskatchewan
53 equipment items found
Manufactured by:NeuroScientific Biopharmaceuticals Ltd. based inCottesloe, AUSTRALIA
Multiple indications with unmet medical need. “Our company goal is to create new products that reach their target in the brain and act as a protector against nerve cells injury. Currently, there are no approved drugs that can slow down or halt neurodegeneration. We believe that EmtinB has potential to bridge this gap and bring this breakthrough medicine to clinics, bringing new hope to ...
Manufactured by:Jointechlabs Inc. based inNorth Barrington, ILLINOIS (USA)
Based on our platform and technology, Jointechlabs is developing therapies for unmet medical needs. Our first therapeutic in clinical development JTL-T-01 is a proprietary injectable stem cell scaffold as a biologic therapy for osteoarthritis, for approval under the FDA’s fast-track program. JTL-T-01 has been designed as an autologous point-of-care therapy ...
Manufactured by:Kangstem Biotech Co., Ltd. based inGangnam-gu, SOUTH KOREA
FURESTEM-AD Inj. is the world’s first stem cell therapeutic product for atopic dermatitis that targets moderate to severe atopic dermatitis ...
Manufactured by:Fate Therapeutics, Inc. based inSan Diego, CALIFORNIA (USA)
The oncology therapeutic landscape has been transformed with the use of checkpoint inhibitor therapy. Despite the clinical benefit conferred by approved checkpoint inhibitor therapy against a variety of tumor types, these therapies are not curative and, in most cases, patients either fail to respond or their disease progresses on these agents. Resistance to checkpoint inhibitor ...
by:Eli Lilly and Company based inIndianapolis, INDIANA (USA)
Our Shielded Living Therapeutics platform combines advanced cell engineering with innovations in biocompatible materials, potentially enabling our products to produce a wide range of therapeutic molecules that may be missing or deficient in the body, including proteins, enzymes or other therapeutic ...
by:Zymeworks Inc based inVancouver, BRITISH COLUMBIA (CANADA)
with a modular suite of proprietary cytotoxins (cell-killing compounds), stable linkers, and conjugation technologies. A broader window of opportunity. Deliver potent cell-killing compounds specifically to diseased target cells with optimal safety and efficacy. ZymeLink™, a next-generation antibody drug conjugate (ADC) platform, is a suite ...
Manufactured by:Pluristem Therapeutics Inc. based inHaifa, ISRAEL
PLX-R18 cells release a combination of therapeutic proteins in response to a damaged or poorly functioning hematopoietic system; this system creates the blood cells that protect us from infection, uncontrolled bleeding and anemia. PLX-R18 is currently in a Phase I clinical trial to treat incomplete hematopoietic recovery following Hematopoietic ...
Manufactured by:Kangstem Biotech Co., Ltd. based inGangnam-gu, SOUTH KOREA
Kangstem Biotech is developing stem cell therapeutic products for patients suffering from rare and incurable diseases using umbilical cord blood-derived stem ...
Manufactured by:Thermo Fisher Scientific based inWaltham, MASSACHUSETTS (USA)
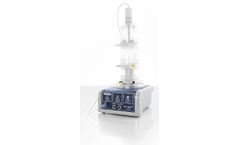
Use the Thermo Scientific™ Nunc™ Automatic Cell Factory™ Manipulator System (ACFM), which features electronic and pneumatic controls, to automate the filling and emptying of a medium or cell suspension into 4 x 3 Nunc Cell Factory 10 systems or 4 x 1 Nunc Cell Factory 40. Specifically designed to lend innovation ...
Manufactured by:Thermo Fisher Scientific based inWaltham, MASSACHUSETTS (USA)
Use the Thermo Scientific™ Nunc™ Automatic Cell Factory™ Manipulator System (ACFM), which features electronic and pneumatic controls, to automate the filling and emptying of a medium or cell suspension into 4 x 3 Nunc Cell Factory 10 systems or 4 x 1 Nunc Cell Factory 40. Specifically designed to lend innovation ...
Manufactured by:Universal Cells Inc. based inSeattle, WASHINGTON (USA)
Realizing the potential of pluripotent stem cells requires that the therapeutic cells derived from PSCs be accepted by patients. PSC-derived cells that have not been engineered to avoid allogeneic rejection will be eliminated soon after transplantation unless they are HLA-matched to their recipients, delivered to immune ...
Manufactured by:Universal Cells Inc. based inSeattle, WASHINGTON (USA)
Pluripotent stem cells (PSCs) such as embryonic stem cells (ESCs) and induced PSCs (iPSCs) are undifferentiated stem cells that can differentiate into any cell type found in the body. Scientists around the world are using PSCs to produce more mature cell types with diverse therapeutic functions. ...
Manufactured by:Regeneron Pharmaceuticals Inc. based inTarrytown, NEW YORK (USA)
elociT™ is our unique mouse technology for producing fully human therapeutic T-cell receptors (TCR) against tumor and viral antigens. Regeneron uses this technology as part of its efforts to discover and develop novel immuno-oncology therapeutic candidates, including combination approaches and to assist our collaborators in producing fully ...
Manufactured by:Human Med AG based inSchwerin, GERMANY
The only system for harvesting regenerative cells (SVF) that can be used at the point-of-care. The new compact Q-graft system for the intraoperative separation and collection of regenerative cells (stromal vascular fraction/ SVF) from adipose tissue will support and enhance the research and clinical application of SVF cells for many ...
Manufactured by:Alphageneron based inCambridge, MASSACHUSETTS (USA)
Chimeric Antigen Receptor (CAR) NK Cells: Alphageneron is developing ‘off-the- shelf’ umbilical cord (UC) blood- and induced pluripotent stem cell (iPSC)-derived CAR-engineered NK (CAR NK) cell-based cancer therapeutics that target tumors expressing mHsp70, and other (undisclosed) targets. Gene editing of allogeneic ...
Manufactured by:RoosterBio, Inc. based inFrederick, MARYLAND (USA)
cGMP xeno-free human bone marrow-derived MSCs to support allogenic therapeutic cell manufacturing. Using a comprehensive and qualified supply chain of our high volume, well-characterized, and standardized WCBs (working cell banks) for large scale cell manufacturing – you’re now able to bypass major steps in cGMP ...
Manufactured by:Terumo Blood and Cell Technologies, Inc. based inLakewood, COLORADO (USA)
The Spectra Optia system is an industry-leading therapeutic apheresis, cell processing and cell collection platform that allows operators to spend more time focusing on patient care. This advanced system uses continuous-flow centrifugation and optical detection technology, providing operators the ability to perform a wide variety of apheresis ...
by:Caribou Biosciences, Inc. based inBerkeley, CALIFORNIA (USA)
Natural killer (NK) cells, as their name implies, play an important role in ridding the body of cancer and viruses. NK cells are emerging as an increasingly important cell type for therapeutic development. Solid tumors are particularly challenging to treat and CAR-T cells have largely underperformed in the ...
Manufactured by:Lineage Cell Therapeutics, Inc. based inCarlsbad, CALIFORNIA (USA)
On December 20, 2021, Lineage announced that it has entered into an exclusive worldwide collaboration and license agreement with Roche and Genentech, a member of the Roche Group, for the development and commercialization of OpRegen for the treatment of ocular disorders, including advanced dry age-related macular degeneration (dry AMD) with geographic atrophy (GA) in a transaction worth up to $670 ...
Manufactured by:Lineage Cell Therapeutics, Inc. based inCarlsbad, CALIFORNIA (USA)
The Lineage Platform begins with an infinitely reproducible cell line. Using our in-house manufacturing abilities and guided differentiation IP, we are able to direct pluripotent cells to become specialized cell types found in the human body, many of which have potential applications in transplant medicine. ...