Show results for
Refine by
Disease Patient Equipment & Supplies In Libya
87 equipment items found
Manufactured by:Baxter International Inc. based inDeerfield, ILLINOIS (USA)
Amia automated peritoneal dialysis (APD) with the Sharesource remote patient management platform is the first and only APD system to include user-friendly features that help guide end-stage renal disease patients through home peritoneal dialysis (PD) therapy, while keeping them remotely connected with their healthcare ...
Manufactured by:Gerresheimer AG based inDüsseldorf, GERMANY
This belt-worn on-body drug delivery device for small-molecule drugs has been developed for our pharma partner EVER Pharma. The wearable device is used by Parkinson's disease patients for the administration of apomorphine to treat this long-term degenerative disorder of the central nervous system. This device is customizable to be used with suitable other drugs ...
Manufactured by:Trukera Medical, formely known as TearLab based inEscondido, CALIFORNIA (USA)
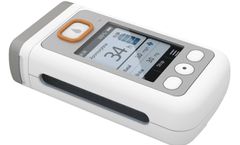
TearLab® Osmolarity System - An Essential Diagnostic for Dry Eye Disease. TearLab is an objective and quantitative point-of-care diagnostic test that provides precise and predictive information. The TearLab Osmolarity System* is intended to measure the osmolarity of human tears to aid in the diagnosis of dry eye disease in patients suspected ...
Manufactured by:MedChemExpress LLC (MCE) based inMonmouth Junction, NEW JERSEY (USA)
Kyotorphin levels in cerebro-spinal fluid correlate negatively with the progression of neurodegeneration in Alzheimer's Disease ...
by:Pureims B.V. based inRoden, NETHERLANDS
Levodopa Cyclops™ is a levodopa dry powder inhaler for the fast and reliable relief of OFF episodes in Parkinson’s disease. As such, Levodopa Cyclops™ answers a major unmet medical need. Levodopa is the cornerstone in the treatment of Parkinson’s disease for well over 50 years. It is the most effective, safe and well tolerated drug ...
Manufactured by:Pharmigene, Inc. based inNeihu Dist, TAIWAN
Clodpgrel, an antiplatelet agents is indicated for the reduction of thrombotic events as follows: Recent MI, Recent Stroke or Established Peripheral Arterial Disease. For patients with a history of recent myocardial infarction (MI), recent stroke, or established peripheral arterial disease, Clopidogrel has been shown to reduce the rate of a ...
Manufactured by:ImpediMed Inc. based inCarlsbad, CALIFORNIA (USA)
SOZO uses ImpediMed’s BIS technology to measure and track critical information about the human body to aid clinicians in managing chronic disease and maximizing patient ...
Manufactured by:ORGENTEC Diagnostika GmbH is part of the Sebia Group based inMainz, GERMANY
The clinical symptoms of APS alone are not sufficiently specific to make a definitive diagnosis. Laboratory tests thus play an important role in the diagnosis of the disease. In patients with APS, autoantibodies are formed that bind to phospholipids like cardiolipin or to phospholipid-binding proteins like beta-2-glycoprotein. Detection of these autoantibodies is ...
Manufactured by:Infitek Co., Ltd. based inJinan, CHINA
Coagulation analyzer is a routine testing medical device that measures the content of various components in human blood, quantifies the results of biochemical analysis, and provides reliable digital basis for clinical diagnosis of various diseases of ...
Manufactured by:Applied Pharma Research s.a. based inBalerna, SWITZERLAND
APR TD012 is a clinical stage drug candidate for the treatment of wounds in Hailey Hailey Disease (HHD) patients. The product improves healing process thanks to its bioburden control and its anti-oxidant properties.Oxidative stress has been demonstrated to play a specific role in the pathogenesis of HHD by regulating the expression of factors playing an ...
Manufactured by:Smith & Nephew plc based in, UNITED KINGDOM
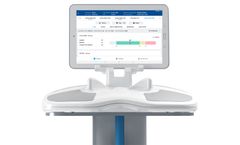
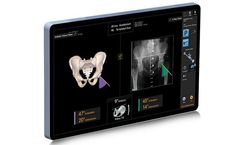
RI.KNEE NAVIGATION puts the surgeon in control of assessing the individual patient disease state and predicting the knee stability over the full range of motion. Navigating gets even better* with a simplified, touch-free interface, pinless options and easy-to-use CLEARLENS ...
by:Reaction Biology Corp. based inMalvern, PENNSYLVANIA (USA)
The Biomarker Analysis tool identifies genes or genetic alterations which are indicative for the efficacy of a drug and which may serve as predictive biomarkers in disease models and patient ...
Manufactured by:OPKO Health, Inc. based inMiami, FLORIDA (USA)
OPK88004 is being assessed as a treatment for muscle wasting, decreased physical strength and function, sarcopenia, and frailty in end stage renal disease (ESRD) patients on dialysis and for prostate cancer patients on Androgen Deprivation Therapy (ADT). The selective anabolic effects of SARM on lean body mass and physical function should provide ...
Manufactured by:TSI Incorporated based inShoreview, MINNESOTA (USA)
To limit the spread of disease through a facility, patients suspected of having diseases spread through the air must be placed in negative pressure rooms, airborne infection isolation rooms. Similarly, immune-compromised patients at risk of contracting diseases should stay in positive pressure, protective ...
Manufactured by:Notal Vision, Inc. based inManassas, VIRGINIA (USA)
Notal Vision is developing a first-of-its-kind Artificial Intelligence-enabled digital diagnostic for patients with neovascular retina diseases using our patient-operated Home Optical Coherence Tomography device. The first disease in the Notal Home OCT Program pipeline is neovascular Age-related Macular Degeneration ...
Manufactured by:MedChemExpress LLC (MCE) based inMonmouth Junction, NEW JERSEY (USA)
Azeliragon (TTP488) is an orally bioavailable inhibitor of the receptor for advanced glycation end products (RAGE) in development as a potential treatment to slow disease progression in patients with mild Alzheimer’s disease (AD)[1]. Azeliragon also can cross the blood-brain barrier ...
Manufactured by:Seres Therapeutics, Inc. based inCambridge, MASSACHUSETTS (USA)
SER-155 is an investigational, oral, rationally designed, fermented microbiome therapeutic designed to reduce the incidence of gastrointestinal infections, bacteremia, and graft versus host disease (GvHD) in immunocompromised patients, including patients receiving allogeneic hematopoietic stem cell transplantation ...
Manufactured by:Entero Therapeutics, Inc. based inBoca Raton, FLORIDA (USA)
Latiglutenase (IMGX003) is an orally administered mixture of two gluten-specific recombinant proteases that degrades gluten proteins into small physiologically irrelevant fragments, and is to be administered as an adjunct to a gluten-free diet (GFD). As illustrated in the below figure, one protease (IMGX001) is effective at breaking glutamine-leucine bonds and the other protease (IMGX002) targets ...
Manufactured by:MGC Diagnostics Corporation based inSaint Paul, MINNESOTA (USA)
Individuals who cannot perform typical spirometry tests, such as the elderly, pediatric or patients with neuromuscular disease are often the patients who need accurate test results the most. That's where the Resmon PRO FULL (V3) comes in. With just normal breathing from the patient, the Resmon PRO FULL (V3) can measure the ...
by:Pure Biologics SA based inWroclaw, POLAND
Myasthenia gravis (MG) is an autoimmune condition classified as a rare disease. Accordingly, myasthenic patients lack effective treatment options, as this rare disease is not a primary interest of the pharmaceutical industry. However, considerable social and institutional support exists for the development of new therapies to treat rare ...