Refine by
Applications
- Microbiome Therapeutics for Patients and Physicians
- Solution for Clinical Trials
- Solution for Access to Investigational Medicines
- Freeze-Dried Hemostatic for Patients
- Drug Dolution for Liver Metastases
- Drug Dolution for Gastric Cancer
- RNA Technology for Research Fellowship
- RNA Technology for Medical Areas of Focus Research
- RNA Technology for Product Pipeline Research
- RNA Technology for Clinical trials
- Radioactive Drugs for Diagnostics
- Radioactive Drugs for Therapeutics
Drug Designation Equipment & Supplies
94 equipment items found
by:Vybion based inMonterey, CALIFORNIA (USA)
INT41 has received Orphan Drug Designation from the FDA for Huntington’s disease. IND enabling studies for INT41 Gene Therapy for Huntington’s disease are in place following a preIND meeting with the FDA in in February 2022 in response to the briefing book prepared by our consultants at Latham BioPharm, a world class consulting group based in ...
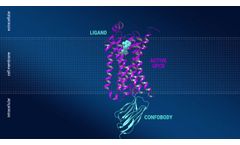
Manufactured by:Confo Therapeutics based inGhent/Zwijnaarde, BELGIUM
With its unique antibody-based technology, Confo Therapeutics aims to overcome the current limitations of GPCR drug discovery in order to fully realize the potential of GPCRs as drug ...
by:Sibylla Biotech S.p.A. based inVerona, ITALY
The PPI-FIT (Pharmacological Protein Inactivation by Folding Intermediate Targeting) technology, of which Sibylla is the exclusive licensee, is a paradigm shift in rational drug design. The basic task of drug design is to find a small molecule able to bind a biological target -such as a protein- so to modulate or block its ...
Manufactured by:Tonix Pharmaceuticals Holding Corp. based inChatham, NEW JERSEY (USA)
TNX-2900, Tonix’s proprietary potentiated intranasal oxytocin is in the pre-Investigational New Drug (IND) stage as a candidate for the treatment of Prader-Willi syndrome (PWS) and non-organic failure to thrive disease. TNX-2900 has been granted Orphan Drug Designation (ODD) in the US by the FDA for the treatment of ...
Manufactured by:Notitia Biotechnologies based inMonmouth Junction, NEW JERSEY (USA)
NM108 is an investigational botanical drug that is designed to treat diseases by promoting the growth of either the existing or replenished Foundation Guild™ bacteria in a patient's gut microbiome. NM108 is currently in phase 2 trial as well as available for Expanded Access (compassionate ...
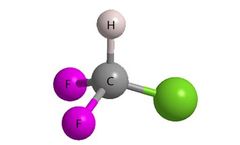
Manufactured by:Alfa Chemistry based inNY, NEW YORK (USA)
Introducing fluorine atom(s) into bioactive organic compounds has become a leading strategy for drug design and lead optimization. In drug synthesis strategy, fluorine is frequently employed to modify biologically relevant properties such as metabolic stability, basicity, lipophilicity and bioavailability. Additionally, the binding affinity of ...
by:Taiwan Liposome Company, Ltd. (TLC) based inNangang Distric, TAIWAN
By encapsulating molecules with poor solubility in polymeric micelle nanoparticles, TLC’s technology can increase the solubility of a drug. This allows a larger quantity to be delivered in a single dose, which can enable new forms of administration. For molecules that are too insoluble to be practical as stand-alone therapies, lipid encapsulation may be the key to ...
Manufactured by:Vivesto AB based inSolna, SWEDEN
Apealea is approved by the European regulatory authority EMA for use in combination with carboplatin for the treatment of adult patients with first relapse of platinum-sensitive epithelial ovarian cancer, primary peritoneal cancer and fallopian tube cancer. Apealea has also received orphan drug designation from the US regulatory authority FDA for the treatment of ...
Manufactured by:Notitia Biotechnologies based inMonmouth Junction, NEW JERSEY (USA)
BM306 is an investigational live biotherapeutic product that contains the Foundation Guild™ bacteria in a defined consortium. This investigational drug is designed to treat diseases by replenishing the missing or extinct Foundation Guild™ bacteria in a patient's gut ...
Manufactured by:SCHOTT Pharma based inMainz, GERMANY
SCHOTT Pharma's cartriQ ready-to-use cartridges are engineered for the safe and effective delivery of peptide- and protein-based drugs through autoinjectors and pen injectors. Constructed from durable FIOLAX® clear Type I Borosilicate Glass, these cartridges offer high resistance to hydrolytic degradation and maintain tight dimensional tolerances for precise dosing. The baked-on ...
Manufactured by:Cellphire Therapeutics, Inc. based inRockville, MARYLAND (USA)
The product is currently in a Phase 2 clinical trial in bleeding thrombocytopenic patients. Thrombosomes has also been given orphan drug designation for the treatment of Acute Radiation Sickness by the U.S. Food and Drug ...
Manufactured by:Rea Tape based in, SOUTH KOREA
THIN GENTLE SILKY FABRICs: REA Tape Premium is made from 100% rayon. LATEX FREE: Is 100% latex free. DRUG FREE: Is 100% free from any medical substances or pharmaceutical drugs. HYPOALLERGENIC: Is designed to minimize the possibility of an allergic response. BREATHABLE: Allows freely for the passage of air and moisture. JAPANESE MEDICAL GLUE: ...
Manufactured by:Rea Tape based in, SOUTH KOREA
100% PURE COTTON: Is made from 100% pure natural cotton fibers. LATEX FREE: Is 100% latex free. DRUG FREE: Is 100% free from any medical substances or pharmaceutical drugs. HYPOALLERGENIC: Is designed to minimize the possibility of an allergic response. BREATHABLE: Allows freely for the passage of air and moisture. JAPANESE MEDICAL GLUE: Uses ...
Manufactured by:Rea Tape based in, SOUTH KOREA
100% PURE COTTON: Is made from 100% pure natural cotton fibers. LATEX FREE: Is 100% latex free. DRUG FREE: Is 100% free from any medical substances or pharmaceutical drugs. HYPOALLERGENIC: Is designed to minimize the possibility of an allergic response. BREATHABLE: Allows freely for the passage of air and moisture. JAPANESE MEDICAL GLUE: Uses ...
Manufactured by:Seres Therapeutics, Inc. based inCambridge, MASSACHUSETTS (USA)
SER-287 is a consortium of multiple bacterial spores manufactured by fractionating and purifying targeted bacteria from stool of healthy human donors. SER-287 has been granted Fast Track designation and Orphan Drug designation by the FDA. A completed SER-287 Phase 1b clinical study demonstrated a statistically significant difference in the ...
Manufactured by:Nemera based inLa Verpillière, FRANCE
Our portfolio of disposable pens includes variable and fixed dose devices for automatic and manual drug administration, combining modern design with high intuitiveness of use for multidose therapies. Nemera offers three disposable pen injector development platforms with wide customization and drug capability options, to serve fulfilling of ...
by:Theramx Inc. based inSeongnam-si, SOUTH KOREA
Drug delivery platform technology designed to maximize efficacy while simultaneously minimizing side effects of existing drugs by delivery to desired ...
Manufactured by:Kala Pharmaceuticals, Inc. based inArlington, MASSACHUSETTS (USA)
AMPPLIFY Drug Delivery Technology is designed to enhance penetration through the mucus barrier and deliver increased concentration of drug to the target ocular tissue. AMPPLIFY Drug Delivery Technology has been shown to improve delivery of loteprednol etabonate compared to traditional suspension eyedrops without ...
Manufactured by:Advin Biotech, Inc. based in, CALIFORNIA (USA)
Our new iSplit Cup and POCiT cup are examples of Advin’s focus on innovative engineering that result in improvements in the drug testing field. These designs remove steps in the lab and/or reduce shipping costs and storage footprint without the need for handling sharps or extra components. Both cups have patents pending and are exclusively available from ...
Manufactured by:Protara Therapeutics, Inc. based inNew York, NEW YORK (USA)
Because PN patients cannot sufficiently absorb adequate levels of choline and no available PN formulations contain sufficient amounts of choline to correct this deficiency, PN patients often experience a prolonged progression to hepatic failure and death, with the only known intervention being a dual small bowel / liver transplant. If approved, IV choline chloride would be the first approved ...