Refine by
Vascular Stent Equipment & Supplies
22 equipment items found
Manufactured by:Veryan Medical Ltd. based inHorsham, UNITED KINGDOM
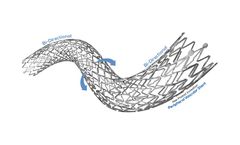
The Challenge: Peripheral Arterial Disease. People are living longer and with an increase in the incidence of obesity, diabetes and other contributing factors, claudication may cause substantial tissue changes and, in some scenarios, can lead to amputation and even death. The peripheral stent market is forecast to grow to $2 billion by 2024 driven by technological advances, an ageing population ...
Manufactured by:Abbott Laboratories based inAbbott Park, ILLINOIS (USA)
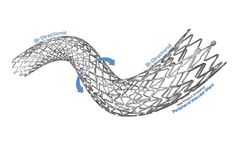
Precision. Performance. Results. Excellent precision, flexibility and proven clinical results in the iliacs. Precision; Triaxial technology designed to absorb stored energy and minimize friction during deployment to ensure precise stent ...
Manufactured by:Stron Medical, Part of Q3 Medical Group based inWinsen, GERMANY
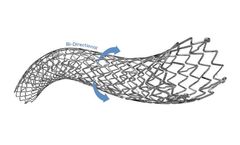
The POLARISADVANCE Peripheral Vascular Self-Expanding Stent System combines highly durable nitinol stent with a flexible delivery system to provide proven clinical ...
Manufactured by:Tsunamed, Part of Q3 Medical Group based inWinsen, GERMANY
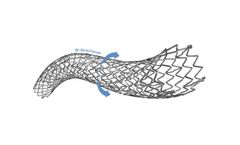
The NAVALISADVANCE Peripheral Vascular Self-Expanding Stent System combines highly durable nitinol stent with a flexible delivery system to provide proven clinical ...
Manufactured by:Tsunamed, Part of Q3 Medical Group based inWinsen, GERMANY
The NAVALIS-ppADVANCE Peripheral Vascular Self-Expanding Stent System combines highly durable nitinol stent with a flexible delivery system to provide proven clinical ...
Manufactured by:Stron Medical, Part of Q3 Medical Group based inWinsen, GERMANY
The POLARIS-ppADVANCE Peripheral Vascular Self-Expanding Stent System combines highly durable nitinol stent with a flexible delivery system to provide proven clinical ...
Manufactured by:pfm Medical Ag based inCologne, GERMANY
The CP Stent is composed of 0.013“ Platinum/Iridium wire that is arranged in a “zig” pattern, laser welded at each joint and over brazed with 24K Gold. It allows expansion from 12 mm to 30 mm. The Covered CP Stent™ is comprised of the Bare CP Stent that is covered with an expandable sleeve of ...
Manufactured by:Terumo Neuro - MicroVention, Inc. based inAliso Viejo, CALIFORNIA (USA)
LVIS and LVIS Jr. devices are braided coil assist stents used for aneurysm ...
Manufactured by:Cirtec Medical based inBrooklyn Park, MINNESOTA (USA)
Vascotube, a Cirtec company, based in Birkenfeld, Germany, processes Nitinol tubing used in implantable medical devices including: Heart Valves, TAVR and TMVR Stents, Peripheral Vascular Stents, Neurovascular ...
Manufactured by:D. E. Hokanson, Inc. based inBellevue, WASHINGTON (USA)
Handheld or pocket Dopplers: the MD6 and the MD6 with Detachable Transducer Dopplers are ideal for locating difficult to find pulses or checking post operative pulses on vascular stents or revascularized sites. ...
Manufactured by:Thoracent, Inc. based inHuntington, NEW YORK (USA)
BONASTENT esophageal stents are among the most technologically advanced non-vascular, self expandable metallic stents available today. Designed with a revolutionary patented, nitinol hook & cross wire structure that allows the stent to adapt and conform to the human anatomy resulting in reduced migration and tumor in-growth. ...
Manufactured by:Artivion, Inc based inKennesaw, GEORGIA (US) (USA)
Based on over 10 years of experience, E-vita OPEN NEO is the next generation hybrid stent graft system for aortic arch and descending aorta repair with Frozen Elephant trunk technique. Each disease requires a unique technique and oversizing strategy, and based on this E-vita OPEN NEO has been created with three stent graft configurations and dedicated designs of ...
Manufactured by:Artivion, Inc based inKennesaw, GEORGIA (US) (USA)
Based on over 10 years of experience, E-vita OPEN NEO is the next generation hybrid stent graft system for aortic arch and descending aorta repair with Frozen Elephant trunk technique. Each disease requires a unique technique and oversizing strategy, and based on this E-vita OPEN NEO has been created with three stent graft configurations and dedicated designs of ...
Manufactured by:Elite Medtek(Jiangsu) Co., Ltd based inChangzhou City, CHINA
The inflation device, specifically utilized in Percutaneous Coronary Intervention (PCI) procedures, is engineered to apply precise pressure to balloon dilation catheters, facilitating the expansion required for vascular enlargements or stent placements. Featuring a quick trigger release mechanism, the device enhances patient safety during operations. Its ...
Manufactured by:Tegra Medical based inFranklin, MASSACHUSETTS (USA)
Highlights: Wire, Tubing, Sheet ...
Manufactured by:CeloNova BioSciences, Inc. based inCarlsbad, CALIFORNIA (USA)
Our proprietary COBRA SG drug-coated PTCA balloon includes a bio-active medical coating designed to improve adhesion of organic and inorganic substrates and optimize drug delivery. Developed at a leading, international medical university, the novel coating enables a unique and efficient excipient to carry a non-proliferative drug on the surface of PTCA balloons and has the flexibility to be ...
Manufactured by:Biosensors International Group, Ltd. based inSingapore, SINGAPORE
In the early 2000's the available mTOR inhibitor limus drugs, available for coronary stenting, were developed for systemic application. Biosensors developed its own proprietary limus technology by focusing during the early stages of drug development on its use for vascular ...
Manufactured by:Becton Dickinson and Company (BD) based inFranklin Lakes, NEW JERSEY (USA)
Tepha is developing fully absorbable stents and coatings made from TephaFLEX® material. This could allow for local drug elution while leaving no permanent residual polymer, which might address the issue of late stent thrombosis with current DESs. In addition, Tepha is developing fully absorbable polymer stents containing TephaFLEX® material, which would provide the necessary support ...
Manufactured by:Bezwada Biomedical LLC based inHillsborough, NEW JERSEY (USA)
Imagine polyurethanes that are completely hydrolysable, biocompatible and have tunable physical and mechanical properties similar to those of commercially available medical grade biostable polyurethanes. At Bezwada Biomedical, we not only imagined it, we have made it into a reality. We are pleased to offer a portfolio of absorbable polyurethanes and their precursors for technical evaluation and ...
Manufactured by:Norman Noble, Inc. based inHighland Hts., OHIO (USA)
Norman Noble’s proprietary STEALTH athermal laser machining provides unmatched machining capabilities on a wide range of ...