Show results for
Refine by
Locations
- USA
- Alabama
- Alaska
- Arizona
- Arkansas
- California
- Colorado
- Connecticut
- Delaware
- District Of Columbia
- Florida
- Georgia (USA)
- Hawaii
- Idaho
- Illinois
- Indiana
- Iowa
- Kansas
- Kentucky
- Louisiana
- Maine
- Maryland
- Massachusetts
- Michigan
- Minnesota
- Mississippi
- Missouri
- Montana
- Nebraska
- Nevada
- New Hampshire
- New Jersey
- New Mexico
- New York
- North Carolina
- North Dakota
- Ohio
- Oklahoma
- Oregon
- Pennsylvania
- Rhode Island
- South Carolina
- South Dakota
- Tennessee
- Texas
- Utah
- Vermont
- Virginia
- Washington
- West Virginia
- Wisconsin
- Wyoming
Covid Kit Equipment Supplied In USA
34 equipment items found
Manufactured by:Co-Diagnostics, Inc. based inSalt Lake City, UTAH (USA)
The Logix Smart™ Coronavirus Disease 2019 (COVID-19) Test kit is an in vitro diagnostic test that uses our patented CoPrimer™ technology for the qualitative detection of the RNA from SARS-CoV-2 coronavirus (COVID-19). The test operates using a single step real-time reverse transcriptase polymerase chain reaction (RT-PCR) process in ...
Manufactured by:Path-Tec, LLC based inMidland, GEORGIA (US) (USA)
Outer box; Outer box label; Medium Prefill; Swab; UN3373 Label on box; 95kPa Biohazard Bag; 50ml Absorbent; Patient Label; UN3373 LabPak; Lot Number ...
by:BioLab Sciences Inc. based inScottsdale, ARIZONA (USA)
BioLab Sciences is distributing Assure COVID-19 test kits. For more information, please click on the buttons below to open up the ...
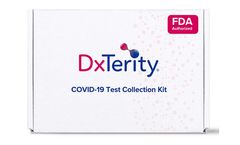
Manufactured by:DxTerity Diagnostics Inc. based inRancho Dominguez, CALIFORNIA (USA)
Our SARS-CoV-2 RT-PCR saliva-based test is FDA EUA authorized for at home unsupervised sample collection, making easy for you to take your test in the comfort of your home. Collect your sample at home and then ship it back to our lab. We use state-of-the-art technology to get your accurate results. Results are reported to you through our secure portal. ...
Manufactured by:NOWDiagnostics Inc. based inSpringdale, ARKANSAS (USA)
The ADEXUSD COVID-19 Test is an in vitro lateral-flow immunoassay intended for qualitative detection of total antibodies to SARS-CoV-2 in human venous whole blood (dipotassium EDTA) and plasma (dipotassium EDTA), serum, and fingerstick whole ...
Manufactured by:Access Bio based inSomerset, NEW JERSEY (USA)
The CareStart™ COVID-19 MDx RT-PCR is a real-time reverse transcription polymerase chain reaction (RT-PCR) test intended for the qualitative detection of nucleic acid from SARS-CoV-2 in respiratory specimens (such as nasopharyngeal, oropharyngeal and nasal swabs, and nasopharyngeal wash/aspirate or nasal aspirate) and bronchoalveolar lavage from individuals suspected of COVID-19 by their ...
Manufactured by:DxTerity Diagnostics Inc. based inRancho Dominguez, CALIFORNIA (USA)
COVID-19 is caused by the SARS-CoV-2 virus which is a new virus in humans causing a contagious respiratory illness. COVID-19 can present with a mild to severe illness, although some people infected with COVID-19 may have no symptoms at all. Older adults and people of any age who have underlying medical conditions have a higher risk of severe illness from COVID-19. Serious outcomes of COVID-19 ...
by:GENETWORx, LLC. based inGlen Allen, VIRGINIA (USA)
GENETWORx is your one stop solution for COVID-19 testing. We now offer COVID-19 nasal swab self-collection test kits, in addition to on-site testing and COVID-19 management software. The GENETWORx COVID-19 Nasal Swab Test Kit enables your employees, students, residents and patients to collect a ...
Manufactured by:Fluxergy based inIrvine, CALIFORNIA (USA)
Learn more about Fluxergy’s qualitative real-time reverse transcription polymerase chain reaction (RT-qPCR) test. For Research Use Only (RUO). Not for use in diagnostic ...
Manufactured by:Fluxergy based inIrvine, CALIFORNIA (USA)
Potential environments for rapid testing applications include emergency rooms, outpatient procedures, urgent care, workplace screening, mobile testing, community level testing settings, school reopenings, and tourism. Fluxergy’s easy-to-use qualitative RT-qPCR test can detect SARS-CoV-2 in one hour. Approved for CE-IVD. All other regions are for research use only. This device is ...
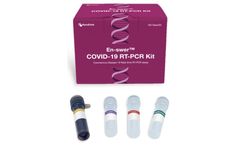
Manufactured by:NanoEnTek, Inc. based inGuro-gu, SOUTH KOREA
The En-swer™ COVID-19 RT-PCR Kit detects E, N, and RNaseP genes of SARS-CoV-2 qualitatively through real-time reverse-transcription polymerase chain reaction. It can be used for rapid detection and outbreak control of COVID-19. ...
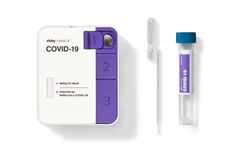
Manufactured by:Visby Medical, Inc. based inSan Jose, CALIFORNIA (USA)
Fast, accurate, and actionable PCR results in under 30 minutes. Instrument-free PCR: Empower on-demand testing, when and where you need ...
Manufactured by:Ellume Limited based inEast Brisbane, AUSTRALIA
Ellume’s swab+drop COVID-19 Self-Swab Kit allows for the self-collection of nasal samples based on easy to follow, animated step-by-step online instructions. The swab+drop Self-Swab Kit has an entry on the Australian Register of Therapeutic Goods (ARTG). We endeavor to work with pathology laboratories to make this product ...
Manufactured by:DxTerity Diagnostics Inc. based inRancho Dominguez, CALIFORNIA (USA)
The DxTerity SARS-CoV-2 (COVID-19) RT-PCR CE saliva-based test provides the convenience of at-home sample collection with results available within 24-72 hours. Our highly-sensitive, gold-standard PCR test has been verified to work with all COVID-19 ...
Distributed by:Alban Scientific, Inc. based inSt. Louis, MISSOURI (USA)
QuickVue At-Home OTC COVID-19 Test lets you get rapid results, in the privacy of your own home. Available over-the-counter, everything you need is in the package and taking the test is simple. The test is authorized for home use with self-collected anterior nasal (nares) swab samples in individuals aged 2 and older. This test is also authorized for home use for individuals aged 2 through 14 ...
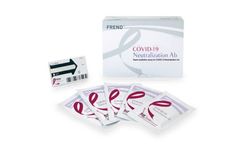
Manufactured by:NanoEnTek, Inc. based inGuro-gu, SOUTH KOREA
The FREND COVID-19 Neutralization Ab is a point-of-care testing (POCT) which can be used to check whether a patient has developed the immune response to SARS-CoV-2 using human serum or plasma ...
Manufactured by:Spark Mobile Diagnostics based inJamnagar, INDIA
CoV-Check DIRECT Covid-19 Antigen Test is a rapid immunochromatographic assay that utilizes specific monoclonal antibodies to detect Nucleocapsid protein of SARS-CoV-2 virus in nasopharyngeal swab specimens. This test provides a quick and easy to use test to help in the diagnosis of SARS-CoV-2 infection to humans. ...
Manufactured by:TBG Diagnostics Limited (TDL) based inMelbourne, AUSTRALIA
The SARS-CoV-2 IgG/IgM Rapid Test Kit is used to qualitatively detect IgG and IgM antibodies of SARS-CoV-2 in human serum, plasma or whole blood. This device is intended to be used by professionals as a screening test and for SARS-CoV-2 research. Any reactive specimen with the SARS-CoV-2 IgG/IgM rapid test kit should be confirmed with alternative testing method(s). ...
Manufactured by:TBG Diagnostics Limited (TDL) based inMelbourne, AUSTRALIA
ExProbe SARS-CoV-2 Testing Kit is used for qualitative detection of the RdRP, N and E genes of SARS-CoV-2 in pharyngeal swabs and sputum samples. The test results of this kit can qualitatively determine whether the suspected patient is infected with 2019 novel coronavirus emerging pneumonia infection of unknown causes. ...
Manufactured by:Access Bio based inSomerset, NEW JERSEY (USA)
Self Test for SARS-CoV-2 Antigen Detection. The CareStart™ COVID-19 Antigen Home Test is a lateral flow immunoassay intended for the qualitative detection of nucleocapsid protein antigens from SARS-CoV-2. Fast and easy to self-test at anywhere. Easy to interpret the results using mobile application. Qualitatively detect the SARS-CoV-2 nucleocapsid protein. Use for nasal swab specimen. Fast ...