Show results for
Refine by
Locations
- USA
- Alabama
- Alaska
- Arizona
- Arkansas
- California
- Colorado
- Connecticut
- Delaware
- District Of Columbia
- Florida
- Georgia (USA)
- Hawaii
- Idaho
- Illinois
- Indiana
- Iowa
- Kansas
- Kentucky
- Louisiana
- Maine
- Maryland
- Massachusetts
- Michigan
- Minnesota
- Mississippi
- Missouri
- Montana
- Nebraska
- Nevada
- New Hampshire
- New Jersey
- New Mexico
- New York
- North Carolina
- North Dakota
- Ohio
- Oklahoma
- Oregon
- Pennsylvania
- Rhode Island
- South Carolina
- South Dakota
- Tennessee
- Texas
- Utah
- Vermont
- Virginia
- Washington
- West Virginia
- Wisconsin
- Wyoming
Medical Sensor Equipment Supplied In USA
25 equipment items found
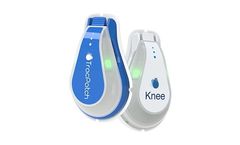
Manufactured by:TracPatch Health based inSacramento, CALIFORNIA (USA)
Monitors knee range of motion, steps, and wound temperature with compact, easy-to-apply sensors that securely uploads real-time data that’s available via the TracPatch apps and web portal, allowing healthcare providers to personalize patients’ recovery journeys throughout the episode of care to optimize the probability of successful ...
Manufactured by:Sensoronics, Inc. based inGig Harbor, WASHINGTON (USA)
OEM Compatible With: Analytical Industries: PSR-11-77; C&S: G0-070; Ceramatec: CAG-17; City Technologies: MOX-4; Datex-Ohmeda: 6051-0000-219; Envitec: OOM105; Hamilton: HM-03; Hudson RCI: V-03, 5558; Integral Process: 65013; Maxtec: MAX-17, R116P10; Mercury Medical: 10-103-02; MSA: 804674; Newport: OM 100, VM 200; Nuova: E-77/O; Sensidyne: SV-03; SensorTech: ST-03; Teledyne: T-7; Ventrex: ...
Manufactured by:Sensoronics, Inc. based inGig Harbor, WASHINGTON (USA)
OEM Compatible With: Analytical Industries: PSR-11-60-03; C&S: G0-07S0; City Technologies: MOX-3; Maxtec: MAX-3 R109P11; Nuova: E-77/OS; Sensidyne: V-08, ...
Manufactured by:Sensoronics, Inc. based inGig Harbor, WASHINGTON (USA)
OEM Compatible With: Analytical Industries: PSR-11-915-2. C&S: G0-020. Draeger: 6850645, 6803290. City Technology: MOX-16. Envitec: OOM201. Datex-Ohmeda: 6051-0000-224. Hamilton: HM-14, HM-13. Hudson-RCI: 5557, V-14. Maxtec: MAX-11, R113P10. Mercury Medical: 10-103-07. MSA: 655263, 655264. Nuova: E-15/2. Teledyne: R-23MED. VTI: ...
Manufactured by:Resonetics based inNashua, NEW HAMPSHIRE (USA)
Resonetics continuously invests in novel technologies to further our expertise, such as in our Electromechanical Sensors for MedTech. We manufacture custom sensors for critical applications in the life ...
Manufactured by:Sensoronics, Inc. based inGig Harbor, WASHINGTON (USA)
OEM Compatible With: Analytical Industries: PSR-11-915-4. C&S: G0-100. Ceramatec: CAG-10. City Technology: MOX-7. Envitec: OOM110. Datex-Ohmeda: 6050-0004-110. Integral Process: 65008. Maxtec: MAX-10, R112P10. Mercury Medical: 10-103-10. Nuova: E-15/4. Pacifitech: PT-20A. Sensidyne: SV-20A. Teledyne: R-30MED. Ventrex: V-20A. Viamed: R-30V. VTI: ...
Manufactured by:Sensoronics, Inc. based inGig Harbor, WASHINGTON (USA)
OEM Compatible With: Air Shields: 6736140; Analytical Industries: PSR-11-55; C&S: G0-130; Draeger: 6736140; Ceramatec: CAG-9; Hudson RCI: V-05, 5500; Integral Process: 65003; Maxtec: MAX-9, R114R80; Mercury Medical: 10-103-03; MSA: 655265; Nuova: E-55/0; Oxitron: 3110; Pacifitech: PT-05; SensorTech: ST-05; Teledyne: R-13; Ventronics: 5500, 5570, 5575, 5584, 5590, 5595; Viamed: ...
Manufactured by:Southland Sensing Ltd. based inOntario, CALIFORNIA (USA)
Intended Use: These oxygen sensors, with their excellent stability, performance and linearity are designed to be used to monitor the partial pressure of oxygen in breathing gas mixtures for anaesthesia, ventilators, medical oxygen concentrators, incubators, and general oxygen ...
Manufactured by:Cubic Sensor and Instrument Co., Ltd based inWuhan, CHINA
Metabolic carbon dioxide (CO2) in the blood is carried to the lungs and exhaled, and EtCO2 levels can reflect the somatic status. Cubic medical mainstream EtCO2 sensor module CM2201 is designed with advanced dual beam non-dispersive infrared technology, and widely used in monitoring the respiratory end tidal CO2 concentration and respiration rate. It can be ...
Manufactured by:Boyd Corporation based inPleasanton, CALIFORNIA (USA)
Wearable Medical Devices, Biosensors, and Monitoring Patches improve patient recovery with increased monitoring of prescribed activities, vitals and other health events. This data combined with new software and artificial intelligence improves diagnosis accuracy and speed as well as increases the efficiency, convenience, customization and cost management of healthcare. Wearable patches and ...
Manufactured by:Lifewave Biomedical, Inc. based inLos Altos, CALIFORNIA (USA)
LifeWave has developed a broad base of intellectual property and expertise in medical radar which uses very low power UWB radio frequency (RF) energy for physiologic assessments. The technology is similar in concept to ultrasound but uses radio frequency instead of sound ...
Manufactured by:Cubic Sensor and Instrument Co., Ltd based inWuhan, CHINA
Metabolic carbon dioxide (CO2) in the blood is carried to the lungs and exhaled, and EtCO2 levels can reflect the somatic status. Cubic medical mainstream EtCO2 sensor module CM2201 is designed with advanced dual beam non-dispersive infrared technology, and widely used in monitoring the respiratory end tidal CO2 concentration and respiration rate. It can be ...
Manufactured by:AII1 Analytical Industries Inc - a brand by Process Sensing Technologies (PST) based inPomona, CALIFORNIA (USA)
USA manufactured and medical certified electrochemical galvanic oxygen sensors with excellent accuracy and stability and individually tested prior to shipment. Fresh sensors direct from manufacturing with extended lifetimes and long warranty. Compliant to FDA, MDSAP, ISO13485, ISO9001 ...
Manufactured by:SM Magnetics, a Quadrant company based inPelham, ALABAMA (USA)
Neodymium Magnets range in BHmax from 28-55 MGOe; Used in almost every industry & application, including medical, motors, consumer electronics, sensors, research, and ...
Manufactured by:Restech based inHouston, TEXAS (USA)
The first and only medical sensor able to monitor real-time, continuous pH in both liquid and aerosolized states, revealing reflux-related etiology of presenting symptoms (e.g. chronic cough, hoarseness, throat clearing, and globus). The sensor is unique in its ability to accurately function anywhere in the aerodigestive tract—from ...
Manufactured by:SSI Electronics, Inc. | Medical Devices based inBelmont, MICHIGAN (USA)
A disposable medical circuit is made up of a variety of components. At a minimum, these devices typically comprise of: a circuit, a patient attachment layer and electronics. In all cases, disposable sensors must adhere directly to a patient’s skin. ...
Manufactured by:Flexpoint Sensor Systems, Inc. based inWest Jordan, UTAH (USA)
The Bend Sensor® is being used in numerous interactive glove applications in both the medical and gaming fields. The sensor gives immediate feedback to hand movement. In November 2009, the Bend Sensor® technology was featured in a study by the University of Rome Tor Vergata, using an interactive glove, and was recommended ...
Manufactured by:Daitron Incorporated based inWilsonville, OREGON (USA)
Daitron Incorporated is a premier manufacturer and distributor of Laser Diode (LD) and Light Emitting Diode (LED) processing solutions. From scribe and break systems to chip and can testers and aging systems, plus visual inspection systems, we manufacturer what you need. We also represent FourTechnos wire bonders and Nikke can ...
by:LOOKEE Tech based inNew York, NEW YORK (USA)
LOOKEE® A310L (Black) Premium Fingertip Pulse Oximeter | Blood Oxygen Saturation Monitor with Alarm, Plethysmograph and Perfusion Index | Finger SpO2 Meter | Batteries, Carrying Case and Lanyard are included. | Approved by FDA and Authorization ID: 326964 in Canada. Aliases: LOOKEE 2022A310LJUN17 | LOOKEE LKA310AL19-BL Care-Kit Device | LOOKEE AAA-BL O-SAT Device | LOOKEE LKEAA-BL Homecare ...
by:LOOKEE Tech based inNew York, NEW YORK (USA)
LOOKEE® A310 (A340) Premium Fingertip Pulse Oximeter | Blood Oxygen Saturation Monitor with Alarm, Plethysmograph and Perfusion Index | Finger SpO2 Tracker | Batteries, Carrying Case and Lanyard are included. | Approved by FDA and Authorization ID: 326964 in Canada. Aliases: LOOKEE 2022A310JUN17 | AAA O-SAT Device | LOOKEE PA3-FPO Tracking Device | LOOKEE LKEAA Homecare Measuring Device | ...