Refine by
Approved Drug Equipment & Supplies
126 equipment items found
by:Curemark based inRye Brook, NEW YORK (USA)
Centers for Disease Control and Prevention estimate that 1 in 68 children in the United States have been diagnosed with Autism. There is currently no approved drug on the market that treats the core symptoms of Autism. At Curemark, we are working to change that. Curemark’s lead drug candidate, CM-AT, has received “Fast Track” ...
by:Curemark based inRye Brook, NEW YORK (USA)
Current prescription medications and psychosocial therapies focus on ameliorating primary symptoms. No currently approved drug treats the underlying cause of ADHD, and there is no known cure. Our pipeline includes a Phase III-ready drug product for ADHD. Preliminary studies with patients diagnosed with ADHD and taking prescribed ADHD medication ...
Manufactured by:Cellworks Research India Private Limited based inBangalore, INDIA
The Cellworks Platform biosimulates how a patient’s unique cancer will respond to therapies prior to treatment and identifies novel drug combinations for treatment-refractory patients. Therapy biosimulation is the ability to predict the phenotype response of a human cell to an external stimulus, such as a drug ligand or radiation. We utilize this ...
Manufactured by:Fate Therapeutics, Inc. based inSan Diego, CALIFORNIA (USA)
In multiple myeloma, malignant plasma cells accumulate in the bone marrow and produce abnormal antibodies called M proteins, which can cause kidney damage, bone destruction, and impaired immune function. While multiple approved drugs with novel mechanisms have improved disease management over the past decade, multiple myeloma is rarely curable and a significant ...
Manufactured by:Vivesto AB based inSolna, SWEDEN
Many intravenously delivered Active Pharmaceutical Ingredients (APIs) are insoluble or poorly soluble in water. This can be a major hurdle in pharmaceutical development and may cause promising drugs to fail during the development process and may limit the application of approved drugs because of poor solubility. According to some estimates, ...
Manufactured by:Cerevast Medical, Inc. based inBothell, WASHINGTON (USA)
The formation or deposition of these clots in the arterial vessels of the brain is one of the primary causes of ischemic stroke. Intravenous thrombolytic therapy (tPA/alteplase, the only approved drug for the treatment of ischemic stroke) works by converting plasminogen in the blood stream to plasmin, the active enzyme that dissolves ...
Manufactured by:AllCells based inAlameda, CALIFORNIA (USA)
AllCells’ GMP Mobilized leukopaks are apheresis products collected from healthy donors treated with FDA-approved drugs to stimulate mobilization of CD34+ hematopoietic stem and progenitor cells from the bone marrow to the peripheral blood. This procedure results in large-scale yields of CD34+ cells. Peripheral blood is collected from IRB-consented donors ...
by:Sibylla Biotech S.p.A. based inVerona, ITALY
In Silico: Identifying small molecules able to revolutionize drug discovery. The PPI-FIT technology allows the exploration of unique pharmacological targets represented by protein folding intermediates. This places Sibylla Biotech in the exclusive position of identifying hit/lead compounds with a mechanism of action never explored so far. ...
Manufactured by:AllCells based inAlameda, CALIFORNIA (USA)
Mobilized Leukopaks are apheresis products collected from healthy donors treated with FDA-approved drugs to stimulate the migration of CD34+ hematopoietic stem and progenitor cells (HSPCs) from the bone marrow niche into the circulation. Mobilized peripheral blood, enriched in single-donor CD34+ HSPCs is collected through leukapheresis using the Spectra ...
Manufactured by:NeuroScientific Biopharmaceuticals Ltd. based inCottesloe, AUSTRALIA
“Our company goal is to create new products that reach their target in the brain and act as a protector against nerve cells injury. Currently, there are no approved drugs that can slow down or halt neurodegeneration. We believe that EmtinB has potential to bridge this gap and bring this breakthrough medicine to clinics, bringing new hope to patients and ...
Manufactured by:MedChemExpress LLC (MCE) based inMonmouth Junction, NEW JERSEY (USA)
N,N-Dimethylacetamide (DMAc) is an organic solvent with blood-brain transmissibility and an FDA-approved drug excipient. N, N-dimethylacetamide exerts anti-inflammatory activity by inhibiting the NF-κB signaling pathway. N, N-dimethylacetamide can be used in studies of weight gain caused by a high-fat diet and neuroinflammation in Alzheimer's ...
Manufactured by:Benevolent based inLondon, UNITED KINGDOM
The emerging global threat from COVID-19 demanded immediate action. With cases rising rapidly, we set up a specialist team to use our AI-drug discovery platform to search for existing drugs — already proven safe — that could be repurposed to treat the virus. We are the only AI company to successfully identify a drug to treat COVID-19. ...
Manufactured by:BioMark Diagnostics Inc. based inRichmond, BRITISH COLUMBIA (CANADA)
BioMark’s initial liquid biopsy assay detects SSAT1 using an FDA approved drug. SSAT1 is an enzyme with elevated levels in numerous cancers. The assay targets a well researched and understood metabolic pathway. BioMark of a few unique companies using safe therapeutic agents for diagnostic indication. As a non-invasive cost effective red alert tool, it is ...
Manufactured by:Beldimed s.a. based inAndrimont, BELGIUM
The expression “Patient First” is part of our DNA. We truly believe that every person in the world should have the same opportunities when it comes to healthcare because health is the ...
Manufactured by:Creative Peptides based inNew York, NEW YORK (USA)
Dalbavancin (INN, trade name Dalvance) is a novel second-generation lipoglycopeptide antibiotic. It belongs to the same class as vancomycin.The Food and Drug Administration approved dalbavancin for the treatment of acute bacterial skin and skin structure infections caused by certain susceptible ...
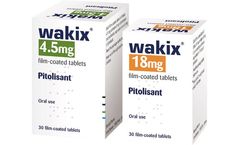
Manufactured by:RareStone Group based inShanghai, CHINA
Pitolisant is a selective histamine H3-receptor antagonist/inverse agonist, which increases the release of the brain chemical histamine to increase a patient's wakefulness and alertness. The drug was approved by the European Medicines Agency (EMA) in 2016 for the treatment of narcolepsy in adults with or with0out cataplexy and is marketed in major countries in ...
Manufactured by:SIGA Technologies, Inc. based inNew York, NEW YORK (USA)
In October 2018, SIGA entered into a Cooperative Research and Development Agreement (CRADA) with the United States Army Medical Research Institute of Infectious Diseases (USAMRIID) with a focus on Good Laboratory Practice (GLP) studies of TPOXX® for PEP using a U.S. Food and Drug Administration (FDA)-approved non-human primate model of TPOXX® efficacy ...
Manufactured by:Eleven P15 based inDenver, COLORADO (USA)
Idiopathic Pulmonary Fibrosis (IPF) is a chronic, fibrosing interstitial pneumonia of unknown cause, occurring primarily in older adults, and localized to the lung. IPF is a fatal disease with median survival at 3-5 years. Two drugs have been approved for treatment of IPF, but these drugs fail to reverse scarred lung tissue or to ...
by:Starton Therapeutics based inParamus, NEW JERSEY (USA)
STAR-LLD is in development in two continuous delivery systems: subcutaneous and transdermal. STAR-LLD is being developed to establish the only IMiD (immunomodulatory drug) approved for CLL. CLL is the most common form of leukemia. Lenalidomide has been shown to be efficacious and well tolerated in CLL previous randomized controlled trials did not obtain an ...
Manufactured by:Amivas (US), LLC based inFrederick, MARYLAND (USA)
Artesunate for Injection is an antimalarial indicated for the initial treatment of severe malaria in adults and ...