Show results for
Refine by
Locations
- USA
- Alabama
- Alaska
- Arizona
- Arkansas
- California
- Colorado
- Connecticut
- Delaware
- District Of Columbia
- Florida
- Georgia (Us)
- Hawaii
- Idaho
- Illinois
- Indiana
- Iowa
- Kansas
- Kentucky
- Louisiana
- Maine
- Maryland
- Massachusetts
- Michigan
- Minnesota
- Mississippi
- Missouri
- Montana
- Nebraska
- Nevada
- New Hampshire
- New Jersey
- New Mexico
- New York
- North Carolina
- North Dakota
- Ohio
- Oklahoma
- Oregon
- Pennsylvania
- Rhode Island
- South Carolina
- South Dakota
- Tennessee
- Texas
- Utah
- Vermont
- Virginia
- Washington
- West Virginia
- Wisconsin
- Wyoming
Covid 19 Automatic Pcr Diatheva Detection Equipment Supplied In Usa
20 equipment items found
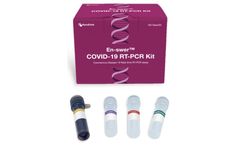
Manufactured by:NanoEnTek, Inc. based inGuro-gu, SOUTH KOREA
The En-swer™ COVID-19 RT-PCR Kit detects E, N, and RNaseP genes of SARS-CoV-2 qualitatively through real-time reverse-transcription polymerase chain reaction. It can be used for rapid detection and outbreak control of COVID-19. ...
Manufactured by:Pathnostics based inIrvine, CALIFORNIA (USA)
The Pathnostics COVID-19 PCR Test detects the presence of SARS-CoV-2, the virus that causes COVID-19. The Pathnostics COVID-19 PCR Test is not impacted by the emerging B.1.1.529 (Omicron variant), enabling accurate test ...
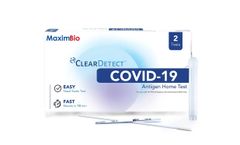
Manufactured by:Maxim Biomedical based inRockville, MARYLAND (USA)
The MaximBio ClearDetect™ COVID-19 Antigen Home Test is an antigen test that allows you to detect proteins from the virus that causes COVID-19. Made easy - An over-the-counter, no prescription needed nasal swab test you can do anywhere & anytime. Know your COVID-19 status in as little as 15 minutes! The MaximBio ClearDetect™ COVID-19 Antigen Home Test is only for use under the ...
Manufactured by:America Diagnostics, Inc. based inSan Diego, CA 92126 USA, CALIFORNIA (USA)
This product is for in vitro diagnostic use, following guidance from the FDA for Emergency Use Authorizations of tests submitted for approval on March 16, 2020. This test has not been reviewed by the FDA and results from antibody testing should not be used as the sole basis to diagnose or exclude SARS-CoV-2 infection or to inform infection ...
Manufactured by:America Diagnostics, Inc. based inSan Diego, CA 92126 USA, CALIFORNIA (USA)
This product is for in vitro diagnostic use, following guidance from the FDA for Emergency Use Authorizations of tests submitted for approval on March 16, 2020. This test has not been reviewed by the FDA and results from antibody testing should not be used as the sole basis to diagnose or exclude SARS-CoV-2 infection or to inform infection ...
Manufactured by:Access Bio based inSomerset, NEW JERSEY (USA)
Self Test for SARS-CoV-2 Antigen Detection. The CareStart™ COVID-19 Antigen Home Test is a lateral flow immunoassay intended for the qualitative detection of nucleocapsid protein antigens from SARS-CoV-2. Fast and easy to self-test at anywhere. Easy to interpret the results using mobile application. Qualitatively detect the SARS-CoV-2 nucleocapsid protein. Use for nasal swab specimen. Fast ...
Manufactured by:Nirmidas Biotech, Inc. based inPalo Alto, CALIFORNIA (USA)
The MidaSpot COVID-19 Antibody Combo Detection Kit is a lateral flow immunoassay intended for the qualitative detection and differentiation of IgM and IgG antibodies to SARS-CoV-2 in human serum, plasma (EDTA or lithium heparin), and capillary whole blood (POC fingerstick) . Nirmidas has obtained FDA EUA for this test.isting fluorescence based microplate ...
Manufactured by:Genabio Diagnostics Inc. based inBedford, MASSACHUSETTS (USA)
cIntended Use: Fosun COVID-19 RT-PCR Detection Kit is a real-time RT-PCR test intended for the qualitative detection of nucleic acid from the SARS-CoV-2 in upper and lower respiratory specimens (such as anterior nasal swabs, mid-turbinate nasal swabs, nasopharyngeal swabs, oropharyngeal swabs, sputum, lower respiratory tract aspirates, bronchoalveolar lavage, and nasopharyngeal wash/aspirate or ...
Manufactured by:Advansta Inc. based inSan Jose, CALIFORNIA (USA)
In December of 2019, the SARS-CoV-2 virus, which causes the disease COVID-19, was first detected in Wuhan, China. The SARS-CoV-2 virus is a single-stranded RNA coronavirus that causes respiratory infections. The rapid spread of COVID-19 has resulted in a global pandemic which has caused an unprecedented social and economic disruption to daily ...
Manufactured by:OPTI Medical Systems, Inc. based inRoswell, GEORGIA (US) (USA)
OPTI Medical Systems has obtained CE marking for its OPTI SARS-CoV-2/Influenza A/B RT-PCR Test for identification and differentiation of SARS-CoV-2, influenza A, and influenza B RNA. The test is available in countries accepting the CE mark. This respiratory panel distinguishes between SARS-CoV-2 and influenza A and B extracted from nasopharyngeal swabs. Use of this test is ideal for identifying ...
Manufactured by:Genscript based inPiscataway, NEW JERSEY (USA)
cPass Neutralization Antibody Detection kit detects circulating neutralizing antibodies against SARS-CoV-2 that block the interaction between the receptor binding domain (RBD) of the viral spike glycoprotein with the ACE2 cell surface receptor. This kit would be instrumental in vaccine and therapeutic development as it is suitable for all antibody isotypes and can be used to determine ...
Manufactured by:SpeeDx based inSydney, AUSTRALIA
Detect COVID-19 causative coronavirus, SARS-CoV-2; Multiplex RT-qPCR test with dual targets (RdRp/ORF1ab) and RNA-based internal control in a single-well. Scalable solution supporting surge ...
Manufactured by:Access Bio based inSomerset, NEW JERSEY (USA)
The CareStart™ COVID-19 Antigen Test is a lateral flow immunochromatographic assay intended for the qualitative detection of the nucleocapsid protein antigen from SARS-CoV-2 in nasopharyngeal or anterior nasal swab specimens directly collected from individuals who are either suspected of COVID-19 by their healthcare provider within the first five days of symptom onset or from individuals ...
Manufactured by:Bio Fire Diagnostics based inSalt Lake City, UTAH (USA)
SARS-CoV-2 is a top concern for patients and clinicians, but several respiratory pathogens can cause nearly indistinguishable symptoms. The FDA De Novo authorized BioFire RP2.1 Panel is a frontline test to help clinicians quickly diagnose respiratory infections, including COVID-19, influenza, RSV, and many ...
Manufactured by:Access Bio based inSomerset, NEW JERSEY (USA)
The CareStart™ COVID-19 MDx RT-PCR is a real-time reverse transcription polymerase chain reaction (RT-PCR) test intended for the qualitative detection of nucleic acid from SARS-CoV-2 in respiratory specimens (such as nasopharyngeal, oropharyngeal and nasal swabs, and nasopharyngeal wash/aspirate or nasal aspirate) and bronchoalveolar lavage from individuals suspected of COVID-19 by their ...
Manufactured by:Co-Diagnostics, Inc. based inSalt Lake City, UTAH (USA)
The Logix Smart™ SARS-CoV-2 DS (Direct Saliva) Test kit is an in vitro diagnostic test that uses our patented CoPrimer™ technology for the qualitative detection of two genes (gene RdRp and gene E) from the RNA of SARS-CoV-2 coronavirus (COVID-19), using a sample processing technique that eliminates the use of the nucleic acid extraction/purification process. ...
Manufactured by:Co-Diagnostics, Inc. based inSalt Lake City, UTAH (USA)
The Logix Smart™ Coronavirus Disease 2019 (COVID-19) Test kit is an in vitro diagnostic test that uses our patented CoPrimer™ technology for the qualitative detection of the RNA from SARS-CoV-2 coronavirus (COVID-19). The test operates using a single step real-time reverse transcriptase polymerase chain reaction (RT-PCR) process in lower respiratory tract fluids (e.g. bronchoalveolar ...
Manufactured by:Co-Diagnostics, Inc. based inSalt Lake City, UTAH (USA)
The Logix Smart™ SARS-CoV-2 (genes RdRp/E) Test kit is an in vitro diagnostic test that uses our patented CoPrimer™ technology for the qualitative detection of two genes (gene RdRp and gene E) from the RNA of SARS-CoV-2 coronavirus (COVID-19). The test operates using a single step real-time reverse transcriptase polymerase chain reaction (RT-PCR) process in lower respiratory tract ...
Manufactured by:Co-Diagnostics, Inc. based inSalt Lake City, UTAH (USA)
The Logix Smart™ ABC (Influenza A/B, SARS-CoV-2) Test kit is an in vitro diagnostic test that uses our patented CoPrimer™ technology for the qualitative detection of the RNA from the Influenza A, Influenza B, and SARS-CoV-2 viruses. The multiplex test operates using a single step real-time reverse transcriptase polymerase chain reaction (RT-PCR) process in lower respiratory tract ...
Manufactured by:Nirmidas Biotech, Inc. based inPalo Alto, CALIFORNIA (USA)
The test can be used for a variety of samples including serum, plasma, whole blood, dry blood spot, and saliva in a lab facility. It can be used to assess IgG and IgM levels in a sample with 100% sensitivity and 99.87% specificity without interferences from other diseases. Avidity pGOLD™ assay can tell if a COVID-19 infection is recent or remote greater than 6 months ...