Refine by
Locations
- USA
- Alabama
- Alaska
- Arizona
- Arkansas
- California
- Colorado
- Connecticut
- Delaware
- District Of Columbia
- Florida
- Georgia (USA)
- Hawaii
- Idaho
- Illinois
- Indiana
- Iowa
- Kansas
- Kentucky
- Louisiana
- Maine
- Maryland
- Massachusetts
- Michigan
- Minnesota
- Mississippi
- Missouri
- Montana
- Nebraska
- Nevada
- New Hampshire
- New Jersey
- New Mexico
- New York
- North Carolina
- North Dakota
- Ohio
- Oklahoma
- Oregon
- Pennsylvania
- Rhode Island
- South Carolina
- South Dakota
- Tennessee
- Texas
- Utah
- Vermont
- Virginia
- Washington
- West Virginia
- Wisconsin
- Wyoming
Coronavirus Testing Equipment Supplied In USA
7 equipment items found
Manufactured by:Seventh Sense Biosystems Inc. based inMedford, MASSACHUSETTS (USA)
Antibodies can help measure your body’s response to vaccination or COVID-19 infection. Use this test to virtually painlessly measure the amount of spike (S) protein antibodies in your blood from the convenience of your home. Currently, only available in ...
Manufactured by:Elabscience based inHouston, TEXAS (USA)
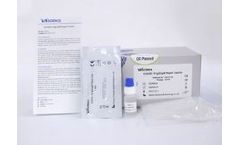
COVID-19 (Corona Virus Disease) is an infectious disease caused by the most recently discovered coronavirus. COVID-19 IgG/IgM Rapid Test ( Serum/Plasma/Whole Blood) is a rapid chromatographic immunoassay for the qualitative of IgG and IgM antibodies to COVID-19 in human serum, plasma or whole ...
by:Thermo Fisher Scientific, LIMS & Laboratory Software based inPhiladelphia, PENNSYLVANIA (USA)
Coronavirus surveillance testing for population-level occurrence. For universities, businesses, and communities eager to reconnect and resume everyday activities, surveillance testing of the SARS-CoV-2 virus is essential to helping monitor population level occurrence. Our PCR testing research solution enables comprehensive, ...
by:T2 Biosystems, Inc. based inLexington, MASSACHUSETTS (USA)
The T2SARS-CoV-2 Panel, run on the T2Dx® Instrument, is a qualitative reverse transcriptase-polymerase chain reaction (RT-PCR) and T2 Magnetic Resonance (T2MR®) test. This test is designed for the direct detection of nucleic acid from SARS-CoV-2 in upper respiratory specimens in transport media from individuals with signs and symptoms of COVID-19. The ...
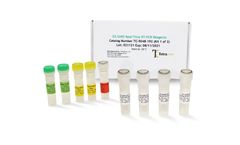
Manufactured by:Tetracore, Inc based inRockville, MARYLAND (USA)
EZ™-SARS-CoV-2 Real-Time RT-PCR is a real-time RT-PCR test intended for the qualitative detection of RNA from the SARS-CoV-2 in human nasal swab specimens from individuals suspected of COVID-19 by their healthcare provider. Testing is limited to laboratories certified under the Clinical Laboratory Improvement Amendments of 1988 (CLIA), 42 U.S.C. §263a, ...
Manufactured by:Co-Diagnostics, Inc. based inSalt Lake City, UTAH (USA)
The Logix Smart™ SARS-CoV-2 (genes RdRp/E) Test kit is an in vitro diagnostic test that uses our patented CoPrimer™ technology for the qualitative detection of two genes (gene RdRp and gene E) from the RNA of SARS-CoV-2 coronavirus (COVID-19). The test operates using a single step real-time reverse transcriptase ...
Manufactured by:Co-Diagnostics, Inc. based inSalt Lake City, UTAH (USA)
The Logix Smart™ Coronavirus Disease 2019 (COVID-19) Test kit is an in vitro diagnostic test that uses our patented CoPrimer™ technology for the qualitative detection of the RNA from SARS-CoV-2 coronavirus (COVID-19). The test operates using a single step real-time reverse transcriptase polymerase ...